Introduction
Isocitrate dehydrogenase 2 (IDH2) is mutated in ~10% of acute myeloid leukemia (AML). However, the clonal evolution of IDH2 mutations through the course of AML has not been clearly elucidated. The presence of targeted therapy, Enasidenib, for the treatment of IDH2 mutated AML underscores the importance of understanding the clonal dynamics of IDH2 mutations.
Methods
IRB approval was obtained. In this study, we analyzed ~6000 patients with NGS results to identify 120 AML patients with IDH2 mutations and longitudinal next generation sequencing (NGS) testing. Disease status was determined for each NGS test date by chart review. IDH2 mutation status was chronicled for each of the following disease states: diagnosis, remission, relapse, and persistent disease. Cytogenetic risk category was based on ELN 2017 guidelines. Statistical analyses were performed using SPSS.
Results
Of the 120 patients, there were 62 patients (51.67%) with AML-NOS and 58 patients (48.33%) with AML with myelodysplasia-related changes (AML-MRC). The most commonly co-occurring mutated genes included DNMT3A, SRSF2, RUNX1, ASXL1, NRAS, BCOR, NPM1, STAG2, FLT3, and PHF6 in order of frequency. Concurrent IDH1 and IDH2 mutations were seen in 2 patients, although IDH1/2 mutations were previously reported to be mutually exclusive.
Of the total patients with IDH2 mutations, 105 patients (88%) were IDH2-positive at the initial diagnosis and 15 patients (12%) were IDH2-negative at diagnosis and acquired the mutation later in disease. Of those 15 patients, 7 patients gained the mutation during persistent disease, 6 during relapse, and 2 at remission (neither of whom relapsed). Forty-eight patients (40%) who were IDH2-positive in a prior test were found to be IDH2-positive with persistent AML, while 11 patients (9%) with IDH2-positive AML lost the IDH2 mutation despite the presence of persistent AML. Twenty-one patients (18%) who were IDH2-positive in a prior test were found to remain IDH2-positive in remission, while 49 patients (41%) cleared the IDH2 mutation. Twenty-four patients (20%) with IDH2-positive AML were found to be IDH2-positive at disease relapse, while 7 patients (6%) lost the IDH2 mutation at relapse.
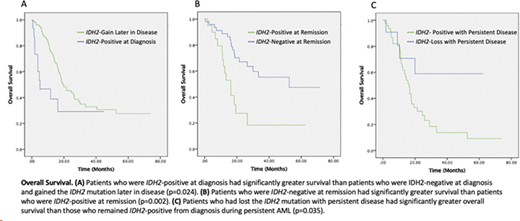
Kaplan-Meier survival analysis and the log-rank test were used to analyze overall survival (OS) to control for confounding factors of AML category (AML-MRC vs AML-NOS) and cytogenetic risk (Figure 1). Patients who were IDH2-positive at diagnosis had significantly better survival than patients who gained the IDH2 mutation later in disease (Figure 1A, p=0.024). Patients who were IDH2-negative at remission had significantly improved survival compared to patients who were IDH2-positive at remission (Figure 1B, p=0.002). Patients who had lost the IDH2 mutation with persistent disease had significantly greater overall survival than those who remained IDH2-positive with persistent AML (Figure 1C, p=0.035). No significant difference in OS was found based on IDH2 mutation status at relapse.
Conclusion
In summary, in the largest study of IDH2 clonal dynamics to date, we found that IDH2 mutations are not stable during AML disease course and frequent genetic testing of AML patients in necessary to tailor personalized therapy. Most patients (70%) cleared IDH2 in disease remission. In those with refractory disease, 18% of IDH2+ AMLs lose IDH2. In the relapse setting, 22% of IDH2+ AML show loss of IDH2. Overall, 12% of patients gained IDH2 mutation later in disease course usually in the setting of refractory/relapsed AML. These patients, along with those who remained IDH2+ in remission and during refractory disease, fared worse than their counterparts. Thus, the longitudinal IDH2 mutation testing at different disease stages may be helpful in prognostic stratification.
Sallman:Agios, Bristol Myers Squibb, Celyad Oncology, Incyte, Intellia Therapeutics, Kite Pharma, Novartis, Syndax: Consultancy; Celgene, Jazz Pharma: Research Funding. Padron:BMS: Research Funding; Novartis: Honoraria; Kura: Research Funding; Incyte: Research Funding. Talati:Pfizer: Honoraria; BMS: Honoraria; Astellas: Speakers Bureau; Jazz: Speakers Bureau; AbbVie: Honoraria. Hussaini:Boston Biomedical: Consultancy; Stemline: Consultancy; Adaptive: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal